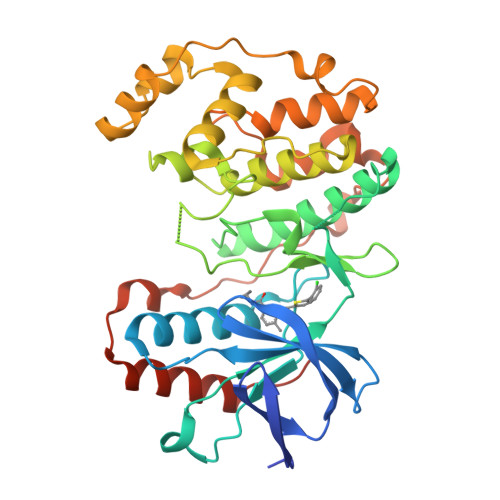
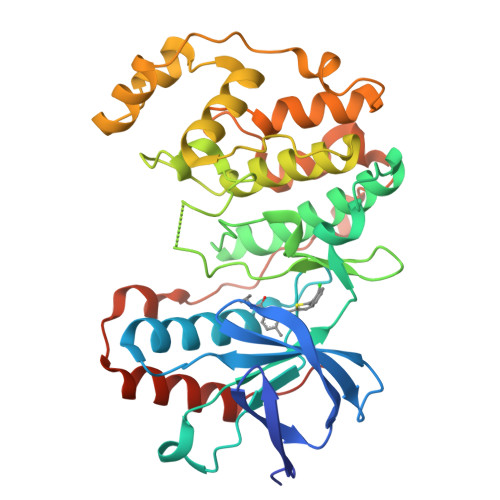
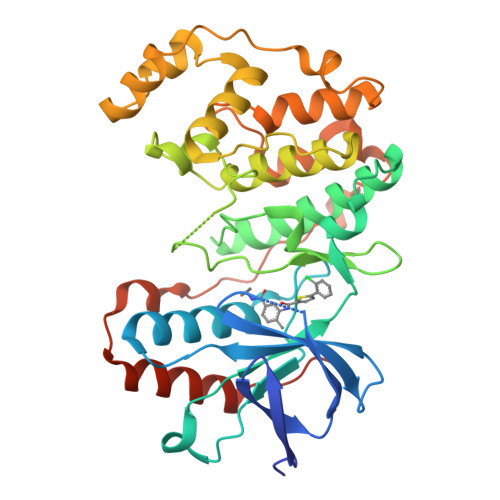
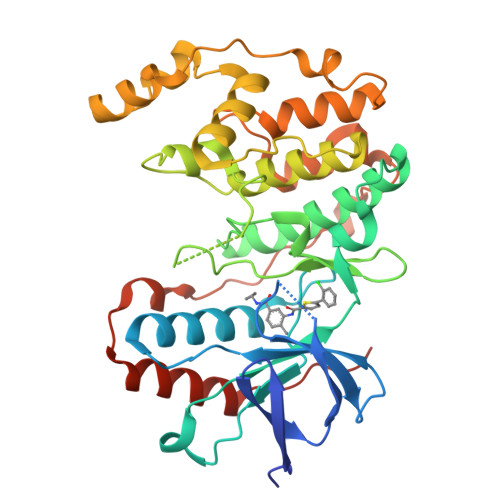
The Identification of Novel P38-Alpha Isoform Selective Kinase Inhibitors Having an Unprecedented P38-Alpha Binding Mode
Wrobleski, S.T., Lin, S., Murali Dhar, T.G., Dyckman, A.J., Li, T., Pitt, S., Zhang, R., Fan, Y., Doweyko, A.M., Tokarski, J.S., Kish, K.F., Kiefer, S.E., Sack, J.S., Newitt, J.A., Witmer, M.R., McKinnon, M., Barrish, J.C., Dodd, J.H., Schieven, G.L., Leftheris, K.(2013) Bioorg Med Chem Lett 23: 4120-4126
- PubMed: 23746475
- DOI: https://doi.org/10.1016/j.bmcl.2013.05.047
- Primary Citation of Related Structures:
4KIN, 4KIP, 4KIQ - PubMed Abstract:
A novel series of p38 MAP kinase inhibitors with high selectivity for the p38α isoform over the other family members including the highly homologous p38β isoform has been identified. X-ray co-crystallographic studies have revealed an unprecedented kinase binding mode in p38α for representative analogs, 5c and 9d, in which a Leu108/Met109 peptide flip occurs within the p38α hinge region. Based on these findings, a general strategy for the rational design of additional promising p38α isoform selective inhibitors by targeting this novel binding mode is proposed.
Organizational Affiliation:
Department of Immunology Chemistry, Bristol-Myers Squibb Pharmaceutical Research Institute, Princeton, NJ 08543-4000, USA. stephen.wrobleski@bms.com